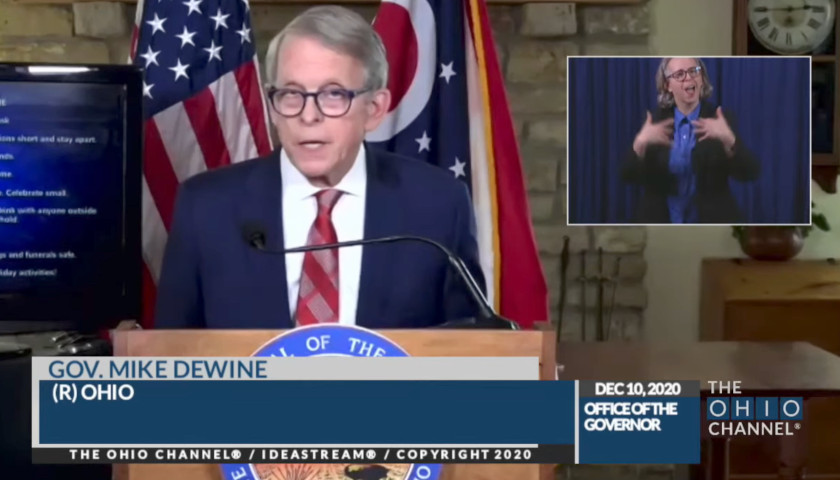
The Ohio Star reported that Ohio Governor Mike DeWine called a special press conference on Monday, November 23 alongside the Ohio Hospital Association leaders to address the state’s COVID hospitalization rise.
As reported, during the briefing doctors who lead each of Ohio’s three zones (the state is segmented into three areas) disclosed staffing shortages due to COVID quarantine orders, which had further depleted caregiving capacity already run thin by upticks in COVID hospital cases around the state.
The Star has received inquiries from readers describing their situations. One woman told the story of her husband who was alerted that he had been exposed to COVID and within days began exhibiting symptoms. When he called his doctor seeking preventative therapies, he was denied. The man was later admitted to a hospital for days, where he received therapeutic treatments that aided his recovery.
Consequently, The Star took the opportunity during Governor DeWine’s twice-weekly COVID presser on Tuesday to ask the following question:
Read More