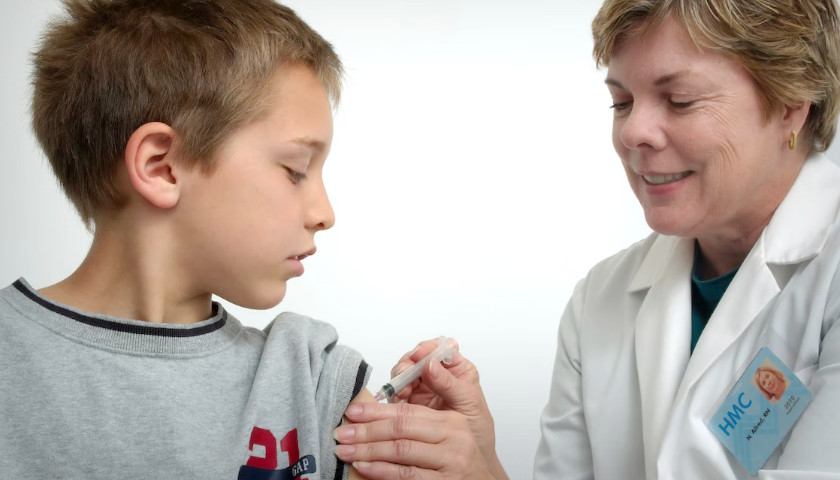
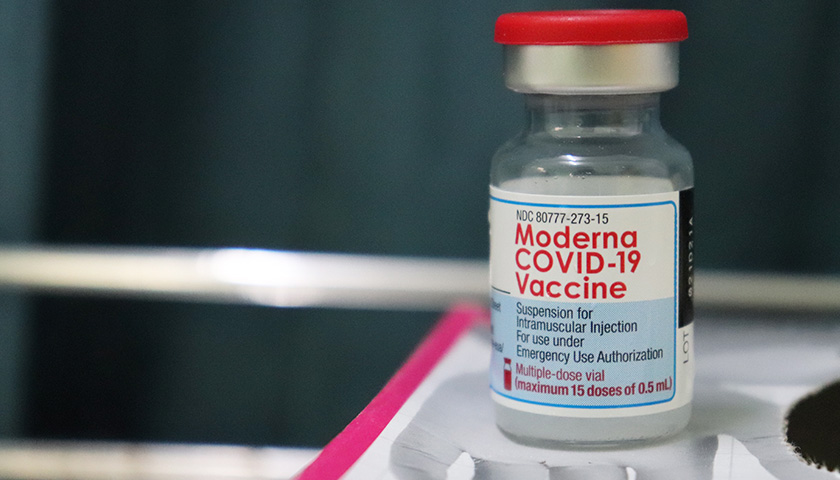
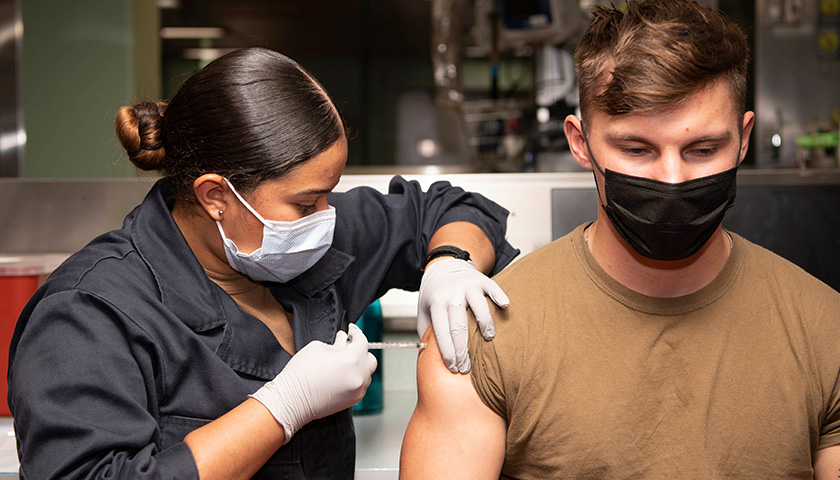
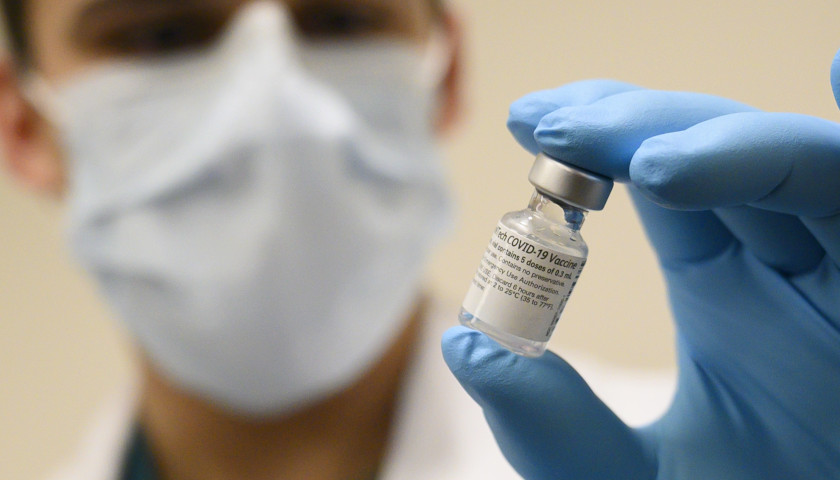
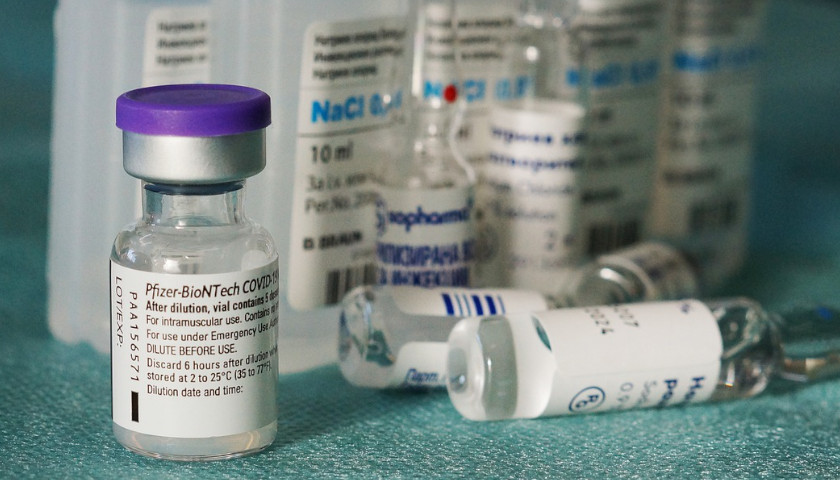
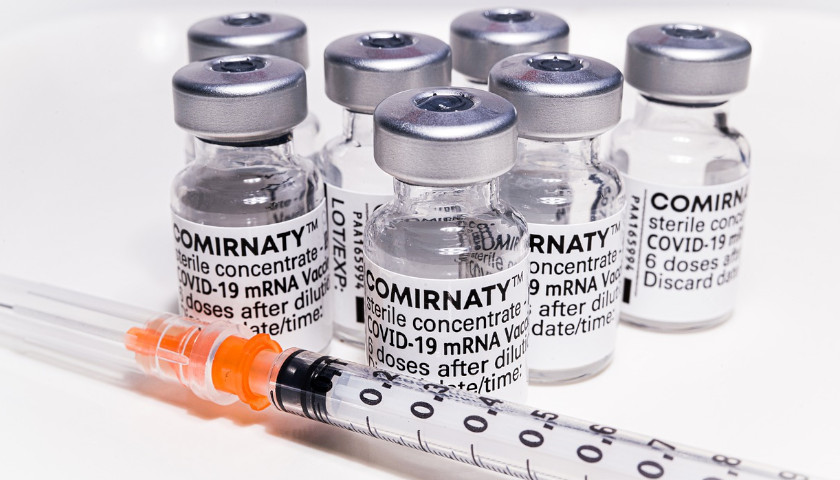
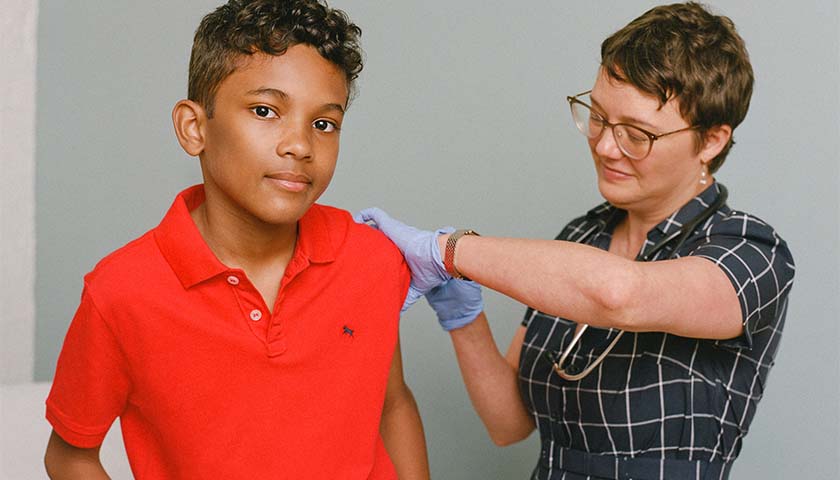
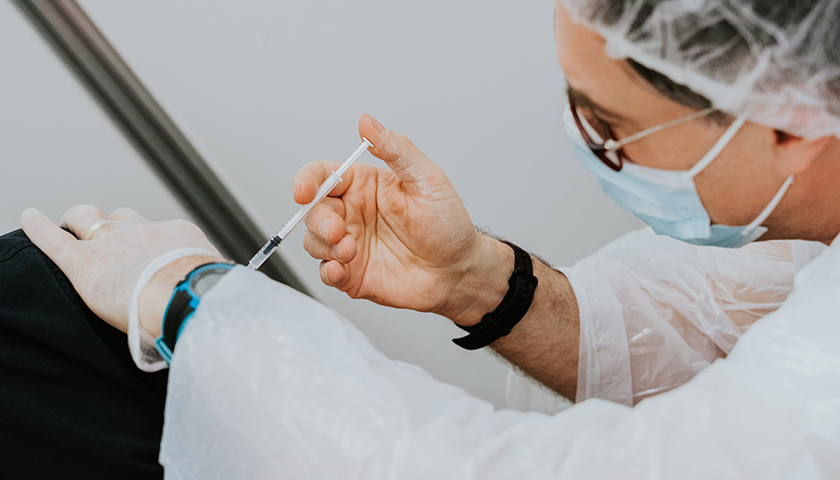
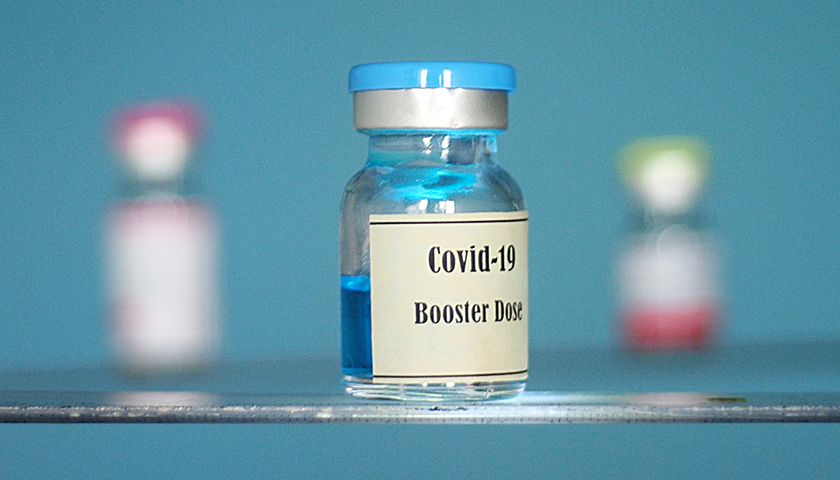
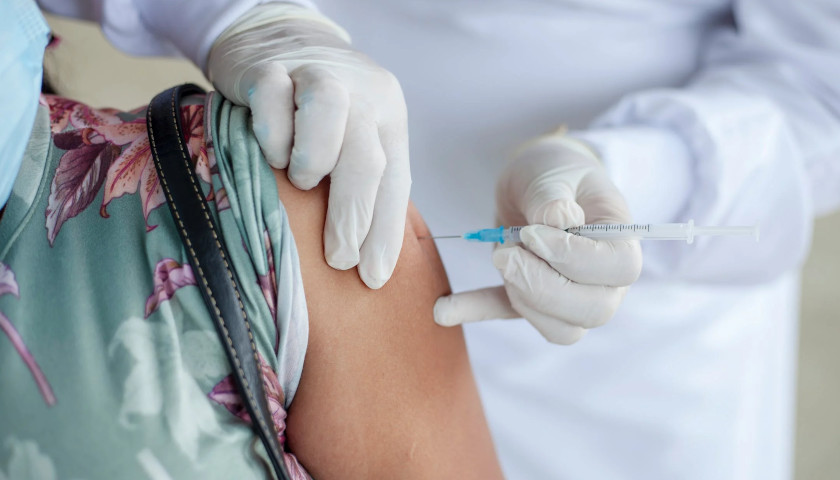
The scientist who first blew the whistle on the DNA contamination in the COVID mRNA injections last year, said Monday that regulators and fact checkers have been “continually wrong” about his alarming discovery, downplaying its significance and telling flat out lies about the potential dangers.
Last April, microbiologist Kevin McKernan, published a paper establishing that simian virus 40 (SV40), a virus found in monkeys and humans, is present in Pfizer and Moderna’s mRNA COVID-19 injections. The discovery was highly significant because SV40 has been linked to cancer in humans, and since the rollout of the mRNA products, the western world has seen a dramatic increase in cancers, especially in previously healthy working aged people.
Read More